A Novel GABA-Based Approach to Major Depressive Disorder (MDD)
In March 2026, ZURZUVAE® (zuranolone) will become available as a novel oral treatment option for major depressive disorder (MDD) and depressive states.
What makes this medication particularly noteworthy is its 14-day treatment course — a short, fixed-duration therapy that may provide rapid symptom improvement compared to traditional antidepressants.
This article explains how zuranolone works, what clinical trials have shown, and what patients should know before considering this new option.
Table of Contents
- What Is Zuranolone?
- Why a 14-Day Treatment Course?
- Mechanism of Action: A GABA-Based Antidepressant
- Phase 3 Clinical Trial Results in Major Depressive Disorder
- Evidence in Postpartum Depression
- Safety and Pharmacokinetic Data
- Side Effects and Clinical Considerations
- Summary
- References
1. What Is Zuranolone?
Zuranolone is a neuroactive steroid and an oral positive allosteric modulator (PAM) of the GABA-A receptor.
Unlike SSRIs and SNRIs, which primarily target serotonin and norepinephrine systems, zuranolone acts on the brain’s inhibitory GABA system, offering a mechanistically distinct approach to treating depression.
It is designed as:
- Once-daily oral dosing
- 14 consecutive days
- A defined short-course treatment strategy
2. Why a 14-Day Treatment Course?
Traditional antidepressants often require 4–6 weeks before significant improvement is observed. This delay can be challenging for individuals experiencing severe depressive symptoms.
Zuranolone was developed to:
- Provide faster onset of antidepressant effects
- Reduce long-term medication burden
- Offer a short-course therapeutic model
Neuroactive steroids directly modulate GABAergic signaling, potentially restoring neural circuit balance more rapidly than monoaminergic agents.
(Cutler AJ, et al., 2023)
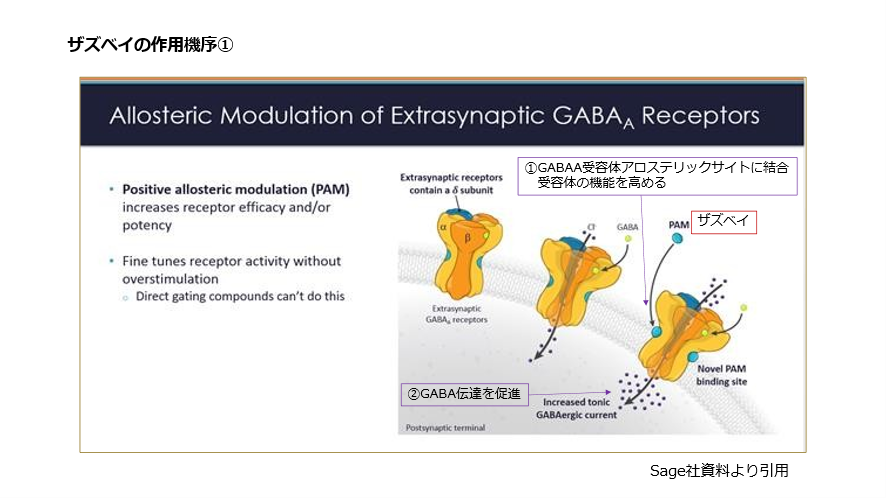
3. Mechanism of Action: A GABA-Based Antidepressant
Zuranolone enhances GABA-A receptor activity as a positive allosteric modulator.
How This Differs from Traditional Antidepressants
| Traditional Antidepressants | Zuranolone |
|---|---|
| Target serotonin/norepinephrine | Targets GABA-A receptors |
| Gradual monoamine modulation | Direct neural inhibition enhancement |
| Often long-term maintenance | 14-day fixed course |
Neuroactive steroids are increasingly recognized for their role in mood regulation and stress-response modulation.
(Cutler AJ, et al., Transl Psychiatry, 2023)
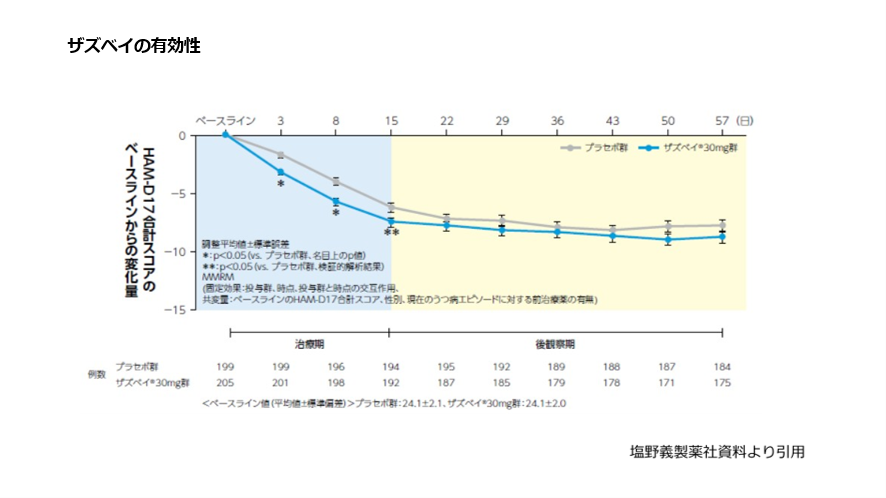
4. Phase 3 Clinical Trial Results in Major Depressive Disorder
A randomized, placebo-controlled Phase 3 trial evaluated zuranolone in adults with major depressive disorder.
Key findings included:
- Significant reduction in HAMD-17 scores compared with placebo
- Improvement observed within days after initiation
- Sustained symptom reduction through Day 15
- Continued benefit beyond treatment completion
(Clayton AH, et al., Am J Psychiatry, 2023)
These results suggest that zuranolone may offer rapid and clinically meaningful improvement in depressive symptoms.
5. Evidence in Postpartum Depression
Zuranolone has also been studied in postpartum depression (PPD).
In a Phase 3 clinical trial, patients with postpartum depression demonstrated:
- Significant symptom improvement compared with placebo
- Rapid onset of antidepressant effects
- Clinically meaningful functional recovery
(Deligiannidis KM, et al., Am J Psychiatry, 2023)
Given the hormonal changes associated with childbirth, the neuroactive steroid mechanism provides a biologically plausible therapeutic approach.
6. Safety and Pharmacokinetic Data
Phase 1 clinical studies evaluated the pharmacokinetics, safety, and tolerability of single and multiple doses of zuranolone in healthy adult participants.
Findings demonstrated:
- A predictable pharmacokinetic profile
- Acceptable tolerability
- No unexpected safety signals
(Sonoyama T, et al., Neuropsychopharmacol Rep, 2023)
These results support its clinical use under appropriate medical supervision.
7. Side Effects and Clinical Considerations
Common adverse effects reported in clinical trials include:
- Somnolence (drowsiness)
- Dizziness
- Headache
- Nausea
Because zuranolone enhances GABAergic inhibition, sedation-related effects may occur.
Patients should exercise caution regarding:
- Driving
- Operating machinery
- Activities requiring full alertness
As with all antidepressants, careful monitoring is recommended during treatment.
8. Summary
ZURZUVAE® (zuranolone) represents a significant advancement in the treatment of major depressive disorder.
Key features include:
- Novel GABA-A receptor mechanism
- 14-day oral treatment course
- Evidence supporting rapid symptom improvement
- Clinical data in both MDD and postpartum depression
- Established safety and pharmacokinetic profile
While not suitable for every patient, zuranolone may provide an important option for individuals seeking a short-course, faster-acting antidepressant strategy.
Consultation with a qualified mental health professional is essential to determine whether this treatment is appropriate.
References
- Cutler AJ, et al. Understanding the mechanism of action and clinical effects of neuroactive steroids and GABAergic compounds in major depressive disorder. Transl Psychiatry. 2023;13:228.
- Clayton AH, et al. Zuranolone for the Treatment of Adults With Major Depressive Disorder: A Randomized, Placebo-Controlled Phase 3 Trial. Am J Psychiatry. 2023;180:676–684.
- Deligiannidis KM, et al. Zuranolone for the Treatment of Postpartum Depression. Am J Psychiatry. 2023;180:668–675.
- Sonoyama T, et al. Pharmacokinetics, safety, and tolerability of single and multiple doses of zuranolone in healthy subjects: A phase 1 clinical trial. Neuropsychopharmacol Rep. 2023;43:346–358.